The Recovered COVID-19 group showed significantly higher numbers of large LDL particles (less atherogenic) than the Acute COVID-19 group (P<0.05). The LDL quality was evaluated using three validated methods: Z-scan, UV-visible spectroscopy, and Lipoprint. Those with positive RT-PCR results at baseline were included in the Acute COVID-19 group (n=33), and those with negative RT-PCR six months after acute infection, were included in the Recovered COVID-19 group (n=30). The participants were adults, aged 18 years or older of both sexes. This study aimed to investigate the atherogenicity (quality) of LDL particles in patients with acute and recovered from COVID-19 infection. 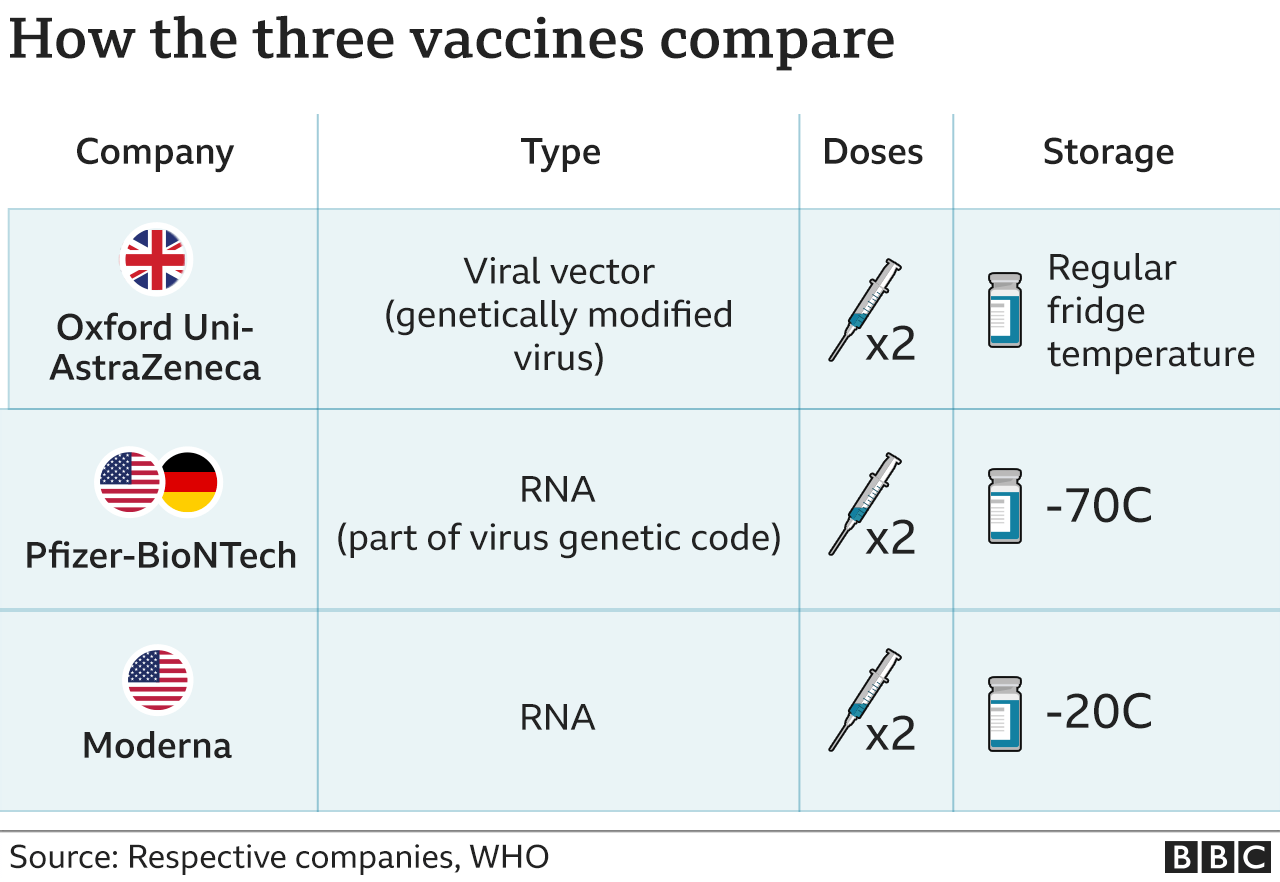
However, it is crucial to consider both the advantages and limitations of these studies and to complement them with experimental validation to ensure the efficacy and safety of identified drug candidates. 
Additionally, in silico studies allow for the prediction of the binding affinity of drug candidates to target receptors, providing insight into their potential efficacy. The use of in silico studies offers several advantages, including the ability to screen a large number of drug candidates in a relatively short amount of time, thereby reducing the time and cost involved in traditional drug discovery methods. In silico studies have identified several promising drug candidates, including Remdesivir, Favipiravir, Ribavirin, Ivermectin, Lopinavir/Ritonavir, and Camostat mesylate, among others. The primary receptors targeted by drug candidates include the RNA polymerase, main protease, spike protein, ACE2 receptor, TMPRSS2, and AP2-associated protein kinase 1. These studies involve the use of computer simulations and computational algorithms to predict the potential interaction of drug candidates with target receptors. In silico studies have emerged as a powerful tool in the drug discovery process, particularly in the search for drug candidates that interact with various SARS-CoV-2 receptors. “Those patients have received e-mails, phone calls, and are also being sent letters to make sure they are aware of this situation.”ĭose recipients were told the only way they could be sure they were fully vaccinated was to get another shot because the doses they’d received had been in the freezer “too long,” an ATC spokesperson said.COVID-19 pandemic has spurred intense research efforts to identify effective treatments for SARS-CoV-2. “While there is no safety risk for the patients, the re-administration is being carried out to ensure that the individuals are fully protected,” a Health Department spokesperson said. New York City’s Health Department communicated with Pfizer the pharmaceutical company said the patients should receive another dose as soon as possible. “After consulting Pfizer, NYC Department of Health sent out an email on our behalf alerting everyone to return for another vaccine.” “We apologize for the inconvenience to those receiving the vaccine batch in question and want people first and foremost to know that we have been advised that there is no danger from the vaccine they received,” the spokesperson said. The doses were given at a pop-up site run by ATC Vaccination Services. 
TIMES SQUARE - Nearly 900 people were administered expired Pfizer COVID vaccine doses at a Times Square site, according to a report. Please look at the time stamp on the story to see when it was last updated. This is an archived article and the information in the article may be outdated. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
